Atorvastatin Related Products
Axios Research stands at the forefront of pharmaceutical reference standards ingenuity, providing high quality Atorvastatin Reference Standards. These include both pharmacopeial and non-pharmacopeial Atorvastatin impurities, metabolites, stable isotope products, and nitrosamines. Our Atorvastatin impurity reference standards are essential for pharmaceutical research, aiding in product development, ANDA and DMF submissions, quality control (QC), method validation, and stability studies. They are also used in identifying unknown impurities and evaluating genotoxic potential. Our Atorvastatin related products are meticulously characterized and come with comprehensive Certificates of Analysis (COA) and analytical data that comply with regulatory standards.
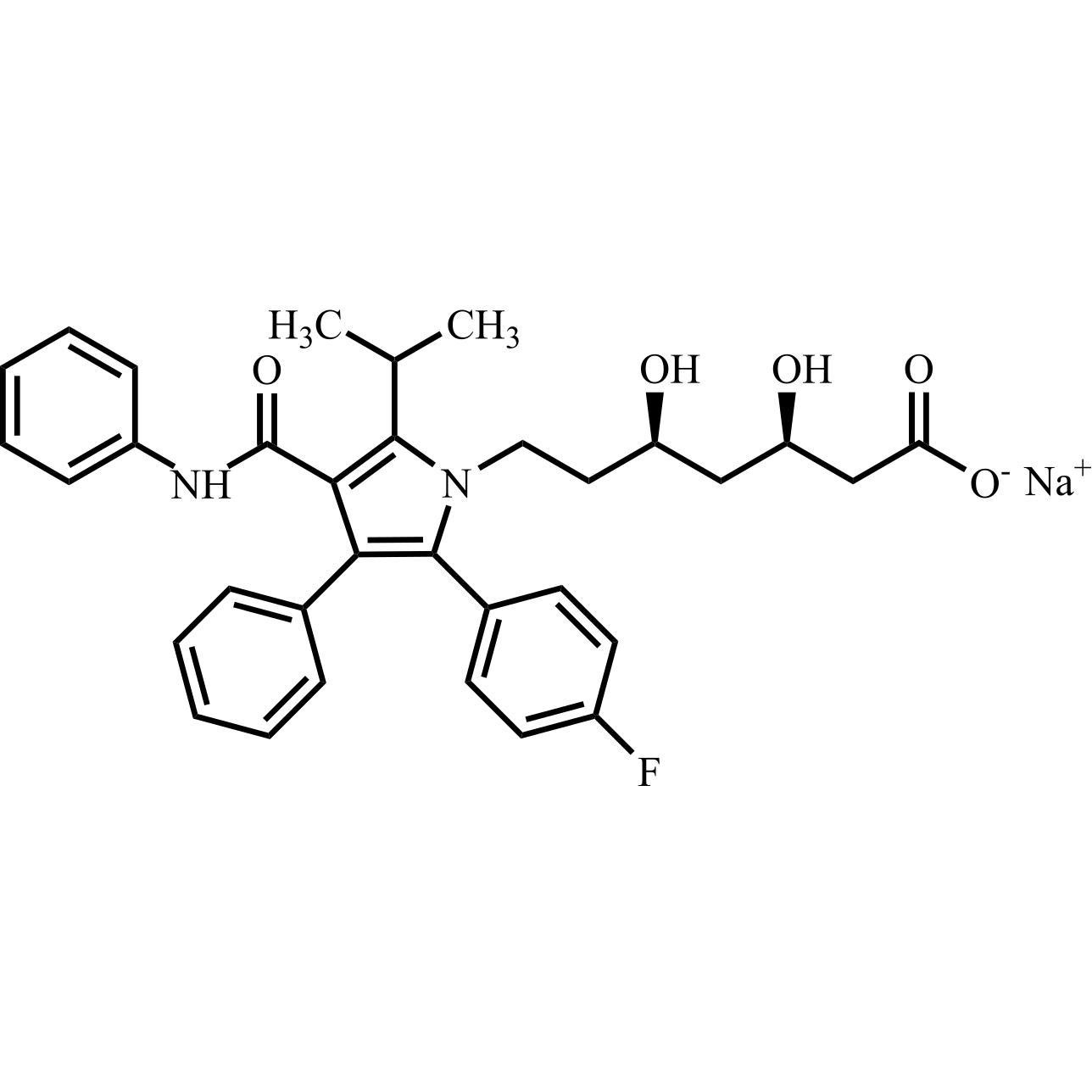
Atorvastatin Calcium Salt
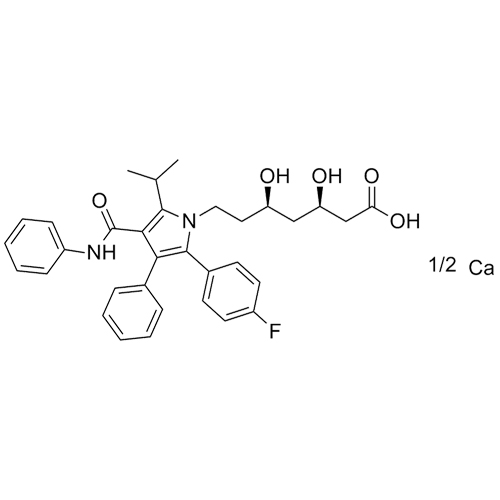
M.F. C₃₃H₃₄FN₂O₅. ₁/₂ Ca
M.W. 557.65 1/2 40.08
CAT# AR-A02771
CAS# 134523-03-8